Dual Use Research of Concern (DURC) is life sciences research that, based on current understanding, can be reasonably anticipated to provide knowledge, information, products, or technologies that could be misapplied to do harm with no, or only minor, modification to pose a significant threat with potential consequences to public health and safety, agricultural crops and other plants, animals, the environment, materiel, or national security.
On May 6, 2025, a new federal policy for oversight of DURC and certain other research with pathogens will go into effect and will apply to all federally funded research (DURC-PEPP Policy). The United States Government Policy for Dual Use Research of Concern and Pathogens with Enhanced Pandemic Potential (“USG DURC-PEPP Policy”) supersedes previous DURC policies and the 2017 Enhanced Potential Pandemic Pathogens Framework (P3CO). The USG DURC-PEPP Policy does not supersede, but complements, other existing federal regulations, including the Select Agent Regulations. The new policy is meant to combine and simplify previous dual-use research and pathogens of pandemic potential policies into a single policy. The intention of the DURC/PEPP policy is to create a culture of biosafety and biosecurity awareness and to institute risk mitigation measures to prevent potential incidents.
Updates to the above policy are expected on or before Tuesday, September 2, 2025 (120 days after the release of a new White House executive order), as communicated in a White House executive order on May 5th, 2025, entitled: Improving The Safety and Security of Biological Research.
Applicability
The DURC-PEPP Policy (and those before it) applies to all investigators at Rutgers University, irrespective of the funding source.
For research to qualify as DURC, it must meet 3 criteria:
- The work must fall within one of the Research Experimental Outcomes
- The work must include a pathogen applicable to the DURC Category for the Research Experimental Outcome(s) identified
- Specific experimental outcomes or actions must be “reasonably anticipated”.
- “Reasonably anticipated” describes an assessment of an outcome such that, generally, individuals with scientific expertise relevant to the research in question would expect this outcome to occur with a non-trivial likelihood. It does not require high confidence that the outcome will definitely occur ,but excludes experiments in which experts would anticipate the outcome to be technically possible, but highly unlikely.
- Principal Investigator (PI) Compliance
- Compliance and cordiality with Rutgers University DURC requirements, the Institutional Contact for Dual Use Research (ICDUR), Institutional Review Entity (IRE), respective funding agencies, and government offices
- Ongoing DURC assessments through grant submissions, IBC submissions, and IRE reviews
- Education and Training of personnel working on the project
- Completion of a Risk: Benefit Analysis and Risk Mitigation Plan related to the identified DURC experiment and pathogen, to the satisfaction of the IRE and funding agency (DURC research specific)
- Progress reports as required by the funding agency and Rutgers (minimally annually for Category 1 and semi-annually for Category 2 (DURC research specific)
It is the responsibility of all Rutgers Principal Investigators to be knowledgeable about and comply with or follow all applicable institutional and U.S. government policies, requirements, and regulations for oversight of biological agent and toxin research.
For PIs and research institutions, failure to follow the USG DURC-PEPP Policy may result in suspension, limitation, or termination of federal funding and loss of future federal funding opportunities for the research proposal and for other life sciences research at the research institution, as imposed by the federal funding agency. Federal funding agencies will consider relevant statutory and regulatory authorities when considering appropriate actions.
- DURC Research Categories
Category 1 Research Category 2 Research Primary Risk The research can be reasonably anticipated to provide, or does provide, knowledge, information, products, or technologies that could be misapplied to do harm with no, or only minor, modification to pose a significant threat with potential consequences to public health and safety, agricultural crops and other plants, animals, the environment, materiel, or national security. Category 1 research may also have biosafety risks. The research can be reasonably anticipated to result in the development, use, or transfer of a pathogen with enhanced pandemic potential (PEPP) or an eradicated or extinct pathogen with pandemic potential (PPP) that may pose a significant threat to public health, the capacity of health systems to function, or national security, through the potential accidental or deliberate introduction of a PEPP or an eradicated or extinct PPP into a human population. Category 2 research may also have dual-use risks. Types of Pathogens in Scope - All Biological Select Agents and Toxins, as listed in 9 CFR 121.3– 121.4, 42 CFR 73.3–73.4, and 7 CFR 331.3, and regulated by USDA and/or HHS
- All Risk Group 4 pathogens listed in Appendix B of the NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules - Classification of Human Etiologic Agents on the Basis of Hazard (NIH Guidelines)
- A subset of Risk Group 3 pathogens listed in Appendix B of the NIH Guidelines3
- For biological agents affecting humans that have not been assigned a Risk Group in the NIH Guidelines, agents affecting humans that are recommended to be handled at Biosafety Level 3 (BSL-3) or Biosafety Level 4 (BSL4) per the BMBL guidance are subject to this Policy.4
- Any pathogen that is modified in such a way that is reasonably anticipated to result in the development, use, or transfer of a PEPP. This includes the development of new PPPs from non-PPPs as well as the enhancement of existing PPPs.
- Eradicated or extinct PPPs that may pose a significant threat to public health, the capacity of health systems to function, or national security
Types of Experimental Outcomes in Scope i. Increase transmissibility of a pathogen within or between host species
ii. Increase the virulence of a pathogen or convey virulence (i.e., the ability of a pathogen to cause disease) to a nonpathogen
iii. Increase the toxicity of a known toxin or produce a novel toxin iv. Increase the stability of a pathogen or toxin in the environment, or increase the ability to disseminate a pathogen or toxin (e.g., improving characteristics of the pathogen or toxin such as environmental stability and ability to be aerosolized) v. Alter the host range or tropism of a pathogen or toxin
vi. Decrease the ability for a human or veterinary pathogen or toxin to be detected using standard diagnostic or analytical methodsvii. Increase resistance of a pathogen or toxin to clinical and/or veterinary prophylactic or therapeutic interventions (e.g., antimicrobials, antivirals, antitoxins, vaccines)
viii. Alter a human or veterinary pathogen or toxin to disrupt the effectiveness of pre-existing immunity, via immunization or natural infection, against the pathogen or toxin
ix. Enhance the susceptibility of a host population to a pathogen or toxini. Enhance transmissibility of the pathogen in humans
ii. Enhance the virulence of the pathogen in humans
iii. Enhance the immune evasion of the pathogen in humans, such as by modifying the pathogen to disrupt the effectiveness of pre-existing immunity via immunization or natural infection
iv. Generate, use, reconstitute, or transfer an eradicated or extinct PPP, or a previously identified PEPPLevel of Review Funding agency review Funding agency and department level review5 PI Progress Report Requirement Annual Semi-annual 3 Note: As of the time of release of this Policy, this subset consists of all RG3 pathogens except HIV, HTLV, SIV, Mtb (including mycobacterium bovis), Clade II of MPVX viruses unless containing nucleic acids coding for clade I MPVX virus virulence factors, vesicular stomatitis virus, Coccidioides immitis, C. posadasii, Histoplasma capsulatum, and H. capsulatum var. duboisii. This list may be updated in the Implementation Guidance on a periodic basis. Beyond this list, as stated in Section 6.2, this Policy also provides voluntary guidance to PIs and research institutions for research that is outside of the scope of this Policy but that may pose a potential risk and may warrant oversight and risk mitigation at the institutional level.
4 Note: In the event no risk group or Biosafety Level has been assigned to an agent, for example in the case of a newly emerging pathogen or chimeric agent, the appropriate institutional body should perform a risk assessment to determine the appropriate Biosafety Level for handling the agent, given the experimental protocol being proposed. The assessment should take into account known properties of the agent and similarities to existing agents. Such agents requiring handling at BSL-3 or BSL-4 are biological agents under Section 4.1.1 of this Policy.
5 In some cases, the federal funding agency and the department funding an in-scope research study are distinct (e.g., the National Institutes of Health and the Department of Health and Human Services). In other cases, they are the same (e.g., the National Science Foundation). Federal departments and agencies will implement this Policy based on their specific departmental structure.- All Biological Select Agents and Toxins, as listed in 9 CFR 121.3– 121.4, 42 CFR 73.3–73.4, and 7 CFR 331.3, and regulated by USDA and/or HHS
- History
DURC policy is an effect of the 2004 Fink report, where seven hazardous experiment classes were initially outlined. On March 29, 2012, the U.S. government released the United States Government Policy for Oversight of Life Sciences Dual Use Research of Concern (2012 Federal DURC Policy). This action established a U.S. government policy for the identification and oversight of DURC. The policy applies to a well-defined subset of life sciences research that involves fifteen agents and toxins and seven categories of experiments (from the 2004 Fink Report) and established regular review of U.S. government-funded or conducted research for its potential to be DURC. On September 24, 2014, the U.S. government released the United States Government Policy for
Institutional Oversight of Life Sciences Dual Use Research of Concern (2014 Institutional DURC Policy). This policy addresses institutional oversight of DURC, including policies, practices, and procedures to identify DURC and implement risk mitigation measures, where applicable.
As a result of certain avian influenza studies in 2013, HHS released an additional oversight framework on a particular subset of potential DURC, known as “gain of function” studies. “Gain-of-Function” studies can help define the fundamental nature of human-pathogen interactions, thereby enabling assessment of the pandemic potential of emerging infectious agents, informing public health and preparedness efforts, and furthering medical countermeasure development. Such studies may entail enhanced biosafety and biosecurity risks; therefore, their risks and benefits must be evaluated, both in the context of recent U.S. biosafety incidents and to keep pace with new technological developments, in order to determine which types of studies should go forward and under what conditions. In October 2014, the U.S. Government Gain-of-Function Deliberative Process and Research Funding Pause on Selected Gain-of-Function Research Involving Influenza, MERS, and SARS Viruses was issued. A deliberative period began when funding for research expected to confer attributes to influenza, MERS, or SARS viruses, such that the viruses would have enhanced pathogenicity and/or transmissibility in mammals via the respiratory route, was paused. During this funding pause, the National Science Advisory Board for Biosecurity (NSABB) was tasked to conduct a deliberative process to draft a set of recommendations for the oversight of gain-of-function research funding that would be reviewed by the broader life sciences community. In 2017, informed by the NSABB recommendations and other inputs, the White House issued the Recommended Policy Guidance for Departmental Development of Review Mechanisms for Potential Pandemic Pathogen Care and Oversight (OSTP P3CO Policy Guidance). Departments and agencies that adopt review mechanisms consistent with the OSTP P3CO Policy Guidance provisions would satisfy the requirements for lifting that department or agency’s funding pause instituted by the U.S. government in 2014. On certain gain-of-function research under the U.S. Government Gain-of-Function Deliberative Process, later in 2017, HHS issued the 2017 HHS P3CO Framework, in accordance with the OSTP P3CO Policy Guidance, lifting the funding pause at HHS and superseding the 2013 HHS HPAI H5N1 Framework. The 2017 HHS P3CO Framework is broader than the 2013 HHS HPAI H5N1 Framework, addressing a wider range of pathogens. The 2017 HHS P3CO Framework and the 2012 and 2014 DURC policies will be unified within the U.S. Government Policy for Oversight of Dual Use Research of Concern and Pathogens with Enhanced Pandemic Potential.
- Purpose
The United States Government Policy for Oversight of Dual Use Research of Concern and Pathogens with Enhanced Pandemic Potential (“Policy”) is a unified federal oversight framework for conducting and managing certain types of federally funded life sciences research on biological agents and toxins. This Policy addresses oversight of research on
biological agents and toxins that, when enhanced, have the potential to pose risks to public health, agriculture, food security, economic security, or national security.1 It supersedes the 2012 United States Government Policy for Oversight of Life Sciences Dual Use Research of Concern (Federal DURC Policy),2 the 2014 United States Government Policy for Institutional Oversight of Life Sciences Dual Use Research of Concern (Institutional DURC Policy),3 and the Recommended Policy Guidance for Departmental Development of Review Mechanisms for Potential Pandemic Pathogen Care and Oversight (P3CO Framework).4 This Policy is issued by the Office of Science and Technology Policy (OSTP) in accordance with the directives established by the 2022 National Biodefense Strategy and Implementation Plan,5 as directed by National Security Memorandum 15,6 to complete an interagency review of efforts to strengthen responsible conduct for biological research. This Policy has also been issued pursuant to Section 2315 of the Consolidated Appropriations Act, 2023 (42 U.S.C. § 6627) to achieve consistent review and oversight of life sciences research proposed for federal funding that may be reasonably anticipated to involve the creation, transfer, or use of pathogens with enhanced pandemic potential (PEPPs).7
1 Risks to national security can arise from, but are not limited to, public health, agriculture, food security, or economic security risks.
2 United States Government Policy for Oversight of Life Sciences Dual Use Research of Concern (2012), https://aspr.hhs.gov/S3/Documents/us-policy-durc-032812.pdf.
3 United States Government Policy for Institutional Oversight of Life Sciences Dual Use Research of Concern (2014), https://aspr.hhs.gov/S3/Documents/durc-policy.pdf.
4 Recommended Policy Guidance for Departmental Development of Review Mechanisms for Potential Pandemic Pathogen Care and Oversight (2017), https://aspr.hhs.gov/S3/Documents/P3CO-FinalGuidanceStatement.pdf.
5 National Biodefense Strategy and Implementation Plan (2022), https://www.whitehouse.gov/wpcontent/uploads/2022/10/National-Biodefens….
6 National Security Memorandum on Countering Biological Threats, Enhancing Pandemic Preparedness, and Achieving Global Health Security (2022), https://www.whitehouse.gov/briefing-room/presidentialactions/2022/10/18….
7 Consistent with 42 U.S.C. § 6627, Category 2 research within this Policy is meant to provide additional oversight and risk mitigation for research with any pathogen that is reasonably anticipated to result in the development, use, or transfer of a PEPP, including the creation of new pathogens with pandemic potential (PPP) from nonPPPs as well as the enhancement of existing PPPs. The United States Government Policy for Oversight of Dual- Rutgers Implementation
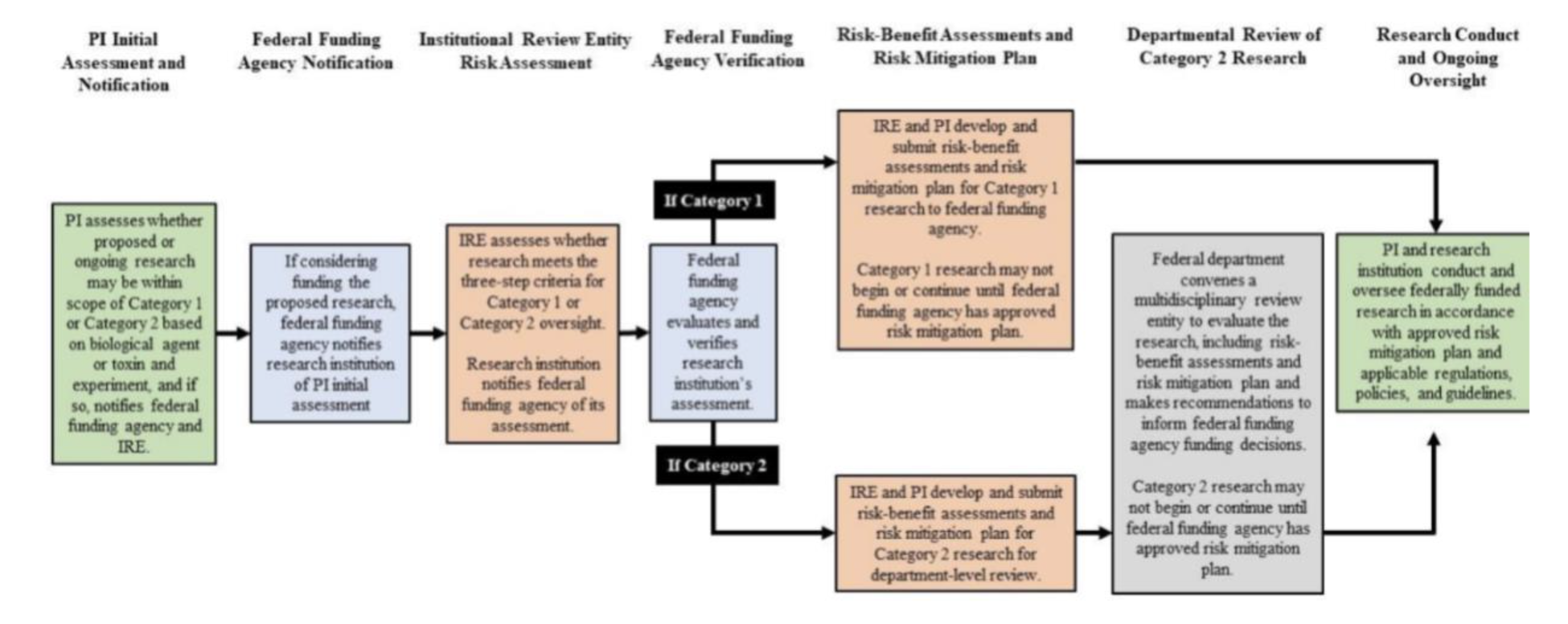
Rutgers University has included DURC as a training topic for biological safety since 2010. It has included the hazardous experiment classes within it in IBC protocol review since 2011 (irrespective of the pathogen being worked with). The IRE will primarily oversee DURC reviews in coordination with the Office of Research and the Biosafety program. Initial DURC review will occur before grant submission to the respective funding agency. Ongoing DURC reviews will be conducted through the IRE and IBC. After receiving IRE and Funding Agency approval (e.g. acceptable Risk: Benefit analysis and Risk Mitigation Plan) IBC approval will be required before the work is allowed to commence per requirements of the NIH Guidelines For Research Involving Recombinant or Synthetic Nucleic Acid Molecules (NIH Guidelines) April 2024.
Rutgers has designated its Institutional Review Entity (IRE) as the Dual Use Research of Concern Committee (DURCom).
IRE Meeting
IRE meeting will be held independently and separately from other institutional committees. Quorum for an IRE meeting will be one representative from each position above for a total of 5 for each IRE review that requires an IRE action. IRE actions are Approve (acceptable to the IRE), Approve with Conditions, Reject (unacceptable to the IRE), Move (time or quorum constraints), and Table (additional information is needed to reach an action). A majority vote is required to allow a submission to proceed in the review process.
IRE Composition
- Committee Chair
- Associate Vice President for Rutgers Research Administration
- Office of General Council Representative to the Office for Research
- Institutional Contact for Dual Use Research (ICDUR)
- Alternate Institutional Contact for Dual Use Research (AICDUR)
- Subject Matter Expert (as required by IRE agenda): Virology, Bacteriology, Mycology, Parasitology, Insects, Animals, Plants, Humans, Transmission, additions can be made at the discretion of the IRE
IRE Review Process
For additional resources, please visit the US Department of Health and Human Services Dual Use Research of Concern or contact durc@research.rutgers.edu.
- Resources
- Improving The Safety and Security of Biological Research
- United States Government Policy for Oversight of Dual Use Research of Concern and Pathogens with Enhanced Pandemic Potential
- Implementation Guidance for the United States Government Policy for Oversight of Dual Use Research of Concern and Pathogens with Enhanced Pandemic Potential
- History of Research Oversight Policies
- Select Agent Regulations
- National Research Council (US) Committee on a New Government-University Partnership for Science and Security. Science and Security in a Post 9/11 World: A Report Based on Regional Discussions Between the Science and Security Communities. Washington (DC): National Academies Press (US); 2007. IV, Biosecurity and Dual-Use Research in the Life Sciences. Available from: www.ncbi.nlm.nih.gov/books/NBK11496
- Press Statement on the NSABB Review of H5N1 Research
- NIH Guidelines For Research Involving Recombinant or Synthetic Nucleic Acid Molecules (NIH Guidelines) April 2024.
General DURC Email: durc@research.rutgers.edu
Committee Chair: Nancy Connell, Ph.D
Institutional Contact for Dual Use Research (ICUDR):
Ryan McAllister Ph.D., M.S., B.S., RBP (ABSA), BRM (IFBA)
Rutgers University Biological Safety Officer, Biosafety Program Manager, and Alternate Responsible OfficialAlternate Institutional Contact for Dual Use Research:
Brian Eggert, MPH, RBP(ABSA)
Senior Biosafety OfficerAlternate Institutional Contact for Dual Use Research:
Jacquelyn Vidal, MS
Assistant Biosafety Officer

